The world is being held hostage by an organism so small that even the most powerful microscope could miss it. It has taken thousands of lives and brought superpowers to their knees. It has also united us in suffering and opened our eyes to the fragility of our way of life. A proposed panacea is vaccination. But will a vaccine be found? Or is the search for one likely to prove fruitless? In order to appreciate the magnitude of such a task, it is important to understand a little of the biology, history and challenges of vaccine development. Here there emerge three good reasons why a vaccine might not be possible. Or, at best, is likely to be a long way off.
Sars-CoV-2 (the virus that gives rise to COVId-19) has an outer membrane which protects the genetic material (RNA) inside. The outer membrane has proteins that help it to invade human cells. One of these proteins, called the spike protein, binds to receptors on the surface of the human lung called ACE2-receptors (see below). Once it binds and enters the cell, it releases its RNA and uses the host’s cell machinery to replicate before invading and damaging adjacent cells.
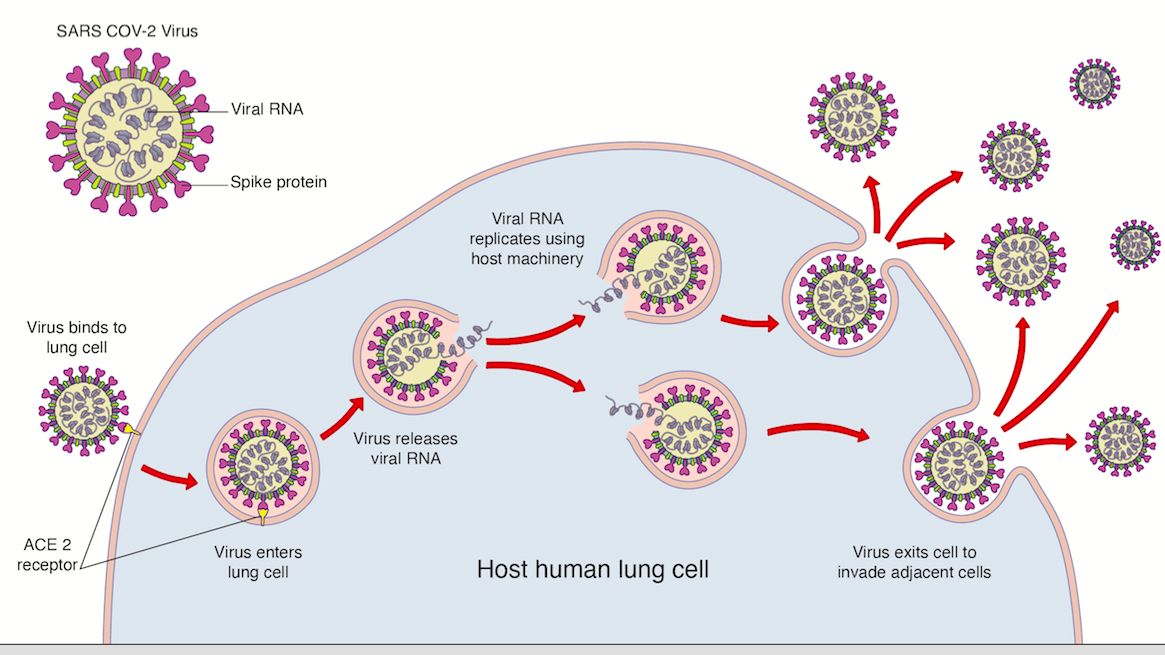
The spike protein on the virus can also be its achilles heel, as it is considered foreign (an antigen) by our body. This means that, once exposed, it is subject to the attack of the body’s immune system which forms the basis of any successful vaccine.
Our immune system is as deadly and as precise as any sophisticated military technology. It can be divided into the first-line, innate response (akin to surface-to-air missiles) and the delayed adaptive response (akin to scrambled F22-Raptor fighter jets). The innate immune system consists of a virus recognition system on cells encountering the virus, which, once activated, releases chemical messengers called cytokines causing an inflammatory cascade to destroy the virus. This buys time for the adaptive response.
The adaptive response is led by two white blood cells: the T-cell (so-called because it matures in the Thymus) and the B-cell (so-called because they were originally discovered in the Bursa (a fluid-filled sac) of birds). One subset of T-cells, called ‘helper T-cells’, recognizes the foreign antigens of the Sars-CoV-2 virus and stimulate B-cells to produce antibodies (immunoglobulins) against it. Immunoglobulins such as IgM and IgG coat the virus and allow it to be eaten by monster cells called phagocytes. Immunoglobulins can also block, for example, the spike protein, rendering it useless; these are called neutralizing antibodies. Another subset of T-cells called ‘cytotoxic T-cells’ kill the virus directly.
A strong immune system is dependent on vitamins, a balanced nutrition and general good health. This is why vitamins such as Vitamin D show promise in defeating the virus. In 80-90 percent of people infected with Sars-CoV-2, this natural response will defeat the viral infection.
However, in a minority of patients, who are typically elderly and have significant co-morbidities, the immune response may be too slow to prevent the virus destroying lung cells. The ensuing lung damage leads to low levels of oxygen in the blood (hypoxemia) which will lead to failure of other organs (kidney, brain etc). These patients will require intensive care treatment in the way of oxygen therapy, non-invasive or invasive ventilation and multi-organ support.
It is particularly in this latter group then that it would be beneficial to develop immunity before the virus causes damage to the body. This is the principle of vaccination. A vaccine is essentially a compound that contains the viral antigens (such as the protein coat of the virus) but not the actual live virus. It fools our immune system into thinking we are infected, thereby mounting the antibody response. These antibodies are kept in the body’s memory so they are primed to kill the virus upon actual infection.
The history of vaccination is fascinating. Edward Jenner in 18th-century England found that milkmaids who had cowpox (a relatively benign disease) didn’t develop smallpox (a comparatively terrible disease). He therefore took the cowpox pustules from a milkmaid, inoculated it into a boy, and then inoculated the boy with smallpox pustules (a research proposal that probably wouldn’t get through a modern ethics committee). The boy didn’t develop smallpox because the antibodies to cowpox worked against smallpox (cross immunity). Progress moved from inoculation of live diluted (attenuated) viruses such as Louis Pasteur’s rabies vaccine to inoculation of dead (inactive) viruses such as polio, which we have today. Jenner’s historic cross immunity may play a part in COVID-19 patients today as there is some evidence that those who have had previous colds may have developed antibodies to the Sars-CoV-2 spike protein which may account for asymptomatic COVID-19 patients.
Vaccination played a significant part in the eradication of smallpox, measles and polio and mandatory immunizations have become routine in most countries. Although the vast majority of vaccines are safe, there has been considerable skepticism of the vaccination industry over the years. Part of the distrust of the industry stemmed from the time the National Childhood Vaccine Injury Act was signed in 1986 which passed liability of compensation claims from alleged vaccine injuries from industry to the federal government.
The race to develop the Sars-CoV-2 vaccine has led to a global framework by institutions such as the European Union to ensure that there is some level of co-operation and sharing of intellectual property to expedite the safe delivery of a vaccine.
However, respiratory vaccines are notoriously difficult to manufacture for three reasons. Firstly, the vaccine immune response may be suboptimal or inappropriate. Suboptimal antibody responses can be corrected to a degree by adding compounds called adjuvants to the vaccine to stimulate a heightened antibody response. Inappropriate or detrimental immune reactions from vaccination are due to a phenomenon called Antibody-Dependent-Enhancement (ADE). ADE had been shown to occur, for example, in persons inoculated with the Respiratory Syncytial Virus (RSV) vaccine in the 1960s where greater hospitalizations were seen in vaccinated children subsequently exposed to RSV. This is why safety assessments of vaccines are vital.
Secondly, the Sars-CoV-2 virus, like influenza, may die away in the summer heat as the transmission rate falls. This means there will not be a great time window to test the vaccine in the population.
Finally, respiratory viruses mutate rapidly. The influenza virus, for example, mutates to such a degree that the antibodies formed by the flu vaccine one year do not recognize the mutant strain the following year.
***
Get three months of The Spectator for just $9.99 — plus a Spectator Parker pen
***
With all these difficulties, it is therefore important to start the development process early. The UK has made a head start in vaccine development. A group at Oxford which has had a respected reputation in developing vaccines has begun human trials for Sars-CoV-2. The British government has invested considerable money to make sure the group has all the resources to develop a vaccine quickly. A similar project in Massachusetts, funded by the National Institute of Health, is also enjoying promising early results and will form part of President Trump’s ‘Warp Speed‘ initiative to develop a vaccine in his trademark expeditious fashion.
However some experts feel that it can take realistically at least two years to develop an effective vaccine due to the reasons outlined above. As well as a researcher, I work as a surgeon. I have operated on COVID-19 patients who have been very sick and it is frightening. Although it is important to mitigate loss of life wherever possible, we can never reduce risk to zero. The majority of people will survive from their natural immunity defenses.
While waiting for a vaccine then, I don’t believe that we should be destroying the income and livelihoods of millions of people, preventing children’s education and social development unnecessarily or canceling treatment for other diseases such as cancer, as this will cause more harm than the virus in the long-term.
Dr Priyad Ariyaratnam is an NIHR clinical lecturer and speciality registrar in cardiothoracic surgery in the Yorkshire & Humber Deanery. This article was originally published on The Spectator’s UK website.